|
3/21/2024 0 Comments Periodic table molar mass cl2This molar mass calculator can only handle two bracket levels at a time. Tricalcium phosphate would be entered as Ca3(PO4)2. Now, compute the sum of products of number of atoms to the atomic weight: Molar mass ( Co (en)Cl 2 Cl) Count i Weight i. Then, lookup atomic weights for each element in periodic table: Cl: 35.453, Co: 58.933195. For example, calcium carbonate would be entered as CaCO3, not caco3. First, compute the number of each atom in Co (en)Cl 2 Cl: Cl: 3, Co: 1. If you know how to calculate molar mass, learn about other ways to express the amount of. Find the molar mass of glucose by multiplying the atomic masses of the atoms and their number, then find the sum: 6 × 12.01 g/mol + 12 × 1.0079 g/mol + 6 × 16 g/mol 180.1548 g/mol. The chemical formula should be entered using standard format. Count the number of atoms of each element in the compound. This calculator is a convenient tool for calculating the molar mass of chemical compounds in lieu of using a periodic table. These include consumption of pH adjustment chemicals for RO feedwater, solubilities of scale forming compounds in reverse osmosis systems, and cation rejection calculations using charge balance (meq/l) in nanofiltration systems. Many other calculations require conversion into moles. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Now, compute the sum of products of number of atoms to the atomic weight: Molar mass (MgCl 2) Count i Weight i. Then, lookup atomic weights for each element in periodic table: Mg: 24.305, Cl: 35.453. Knowing the desired concentration of ClO2, the system integrator can calculate the consumption of each of the reactants using the stoichiometric relationship:ĢNaClO2 + NaOCl + 2HCl ↔ 2ClO2 + H2O + 3NaCl Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.The chemical symbol for Hydrogen is H. First, compute the number of each atom in MgCl 2: Mg: 1, Cl: 2. Now, compute the sum of products of number of atoms to the atomic weight: Molar mass (2MgCl 2) Count i Weight i. For example, certain types of chlorine dioxide (ClO2) generators would use sodium hypochlorite (NaOCl), sodium chlorite (NaClO2) and hydrochloric acid (HCl). First, compute the number of each atom in 2MgCl 2: Mg: 2, Cl: 4. Thus, the molar mass of bilirubin can be expressed as 584.73 g/mol, which is read as “five hundred eighty four point seventy three grams per mole.When calculating consumption of certain RO chemicals for reverse osmosis pretreatment or post-treatment, it is often necessary to convert into moles. The division sign (/) implies “per,” and “1” is implied in the denominator. For example, the molar mass of Ba(OH) 2 requires the sum of 1 mass of Ba, 2 masses of O, and 2 masses of H: The molar mass of Ba(OH)2 requires the sum of 1 mass of Ba, 2 masses of O, and 2 masses of H: 1 Ba molar mass:īecause molar mass is defined as the mass for 1 mol of a substance, we can refer to molar mass as grams per mole (g/mol).  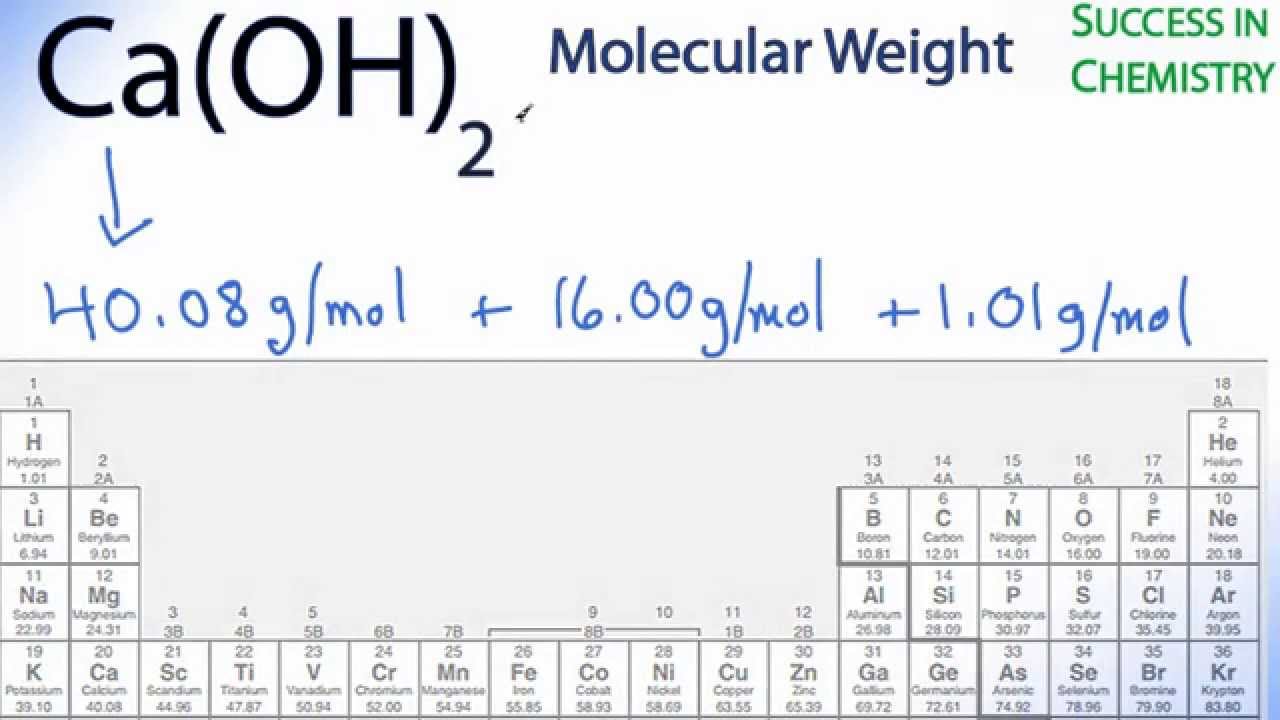
In formulas with polyatomic ions in parentheses, the subscript outside the parentheses is applied to every atom inside the parentheses. 
adenosine (C 10H 13N 5O 4), a component of cell nuclei crucial for cell divisionīe careful when counting atoms.barium sulfate (BaSO 4), used to take X rays of the gastrointestional tract. 
The calculator takes the elemental composition of the compound and weighs the elements to get an empirical formula mass. What is the mass of 1 mol of each substance? Our molar mass calculator uses the periodic table and the chemical formula to solve for the molar mass of a chemical compound based on the compounds empirical formula. \): Moles to Mass Conversion with Compounds
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
